Immune Profiling
Maxpar Direct Immune Profiling Assay and Maxpar Pathsetter software
Introducing the first complete sample-to-answer solution for single-cell high-dimensional immune profiling of human PBMC and whole blood, the Maxpar® Direct™ Immune Profiling Assay™ and Maxpar Pathsetter™ software. Each provides the data quality and reproducibility required to capture the highly individual nature of immune response in health and disease.
No CyTOF? No problem.
We can help you convert your fluorescence panel and find a service provider.

The technical details: populations, clones and workflows
Designed as a simple single-tube workflow, the system brings together CyTOF® technology on the CyTOF XT™ and Helios™ mass cytometry systems, a dry-format 30-marker antibody panel and automated Maxpar Pathsetter software, enabling identification of 37 immune cell populations.
Get more details from the Maxpar Direct Immune Profiling flyer.

Single-tube antibody panel design. All antibody clones used in the kit are familiar and industry-proven.
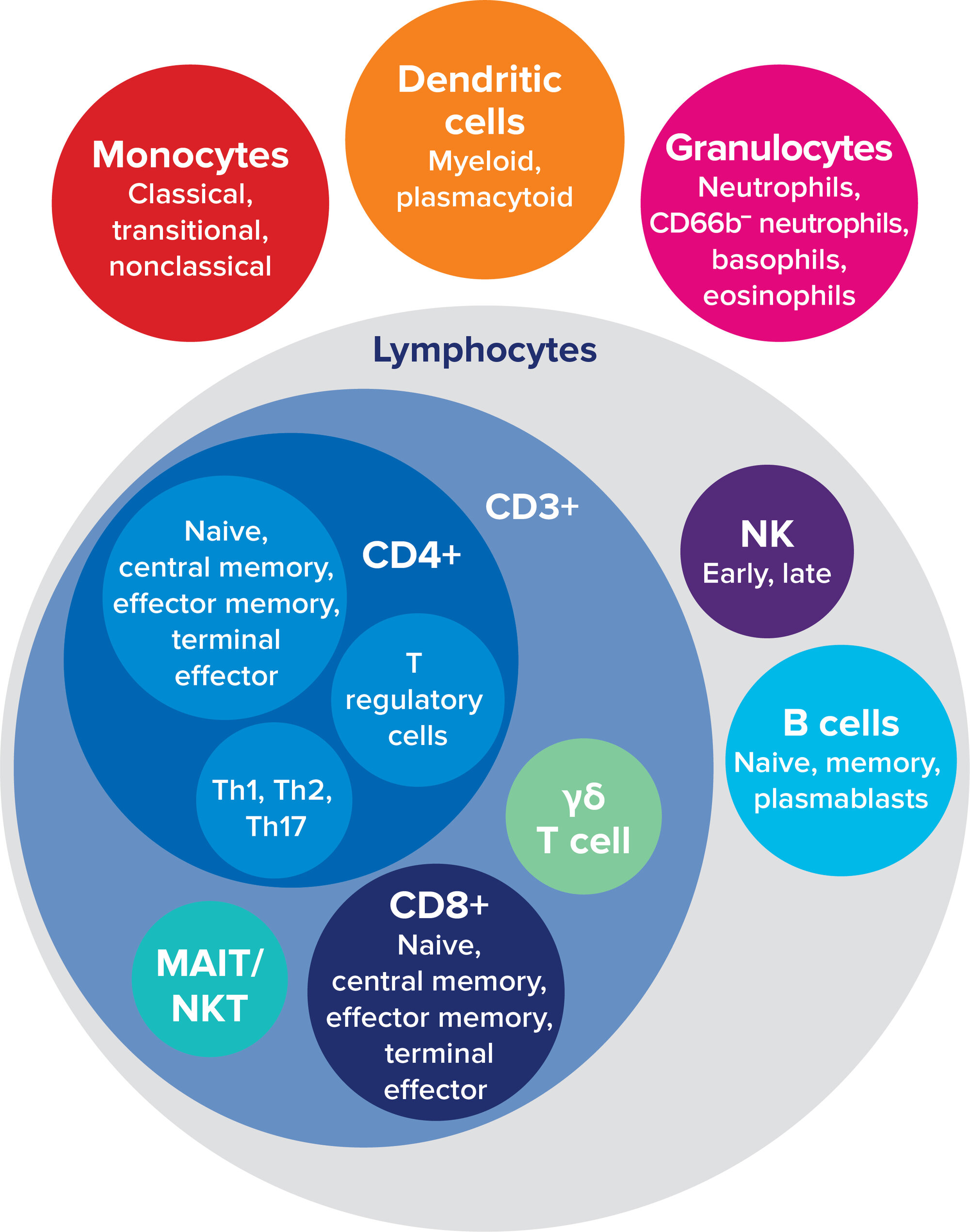
The 37 immune cell subsets identified using the Maxpar Direct Immune Profiling Assay and Maxpar Pathsetter software.
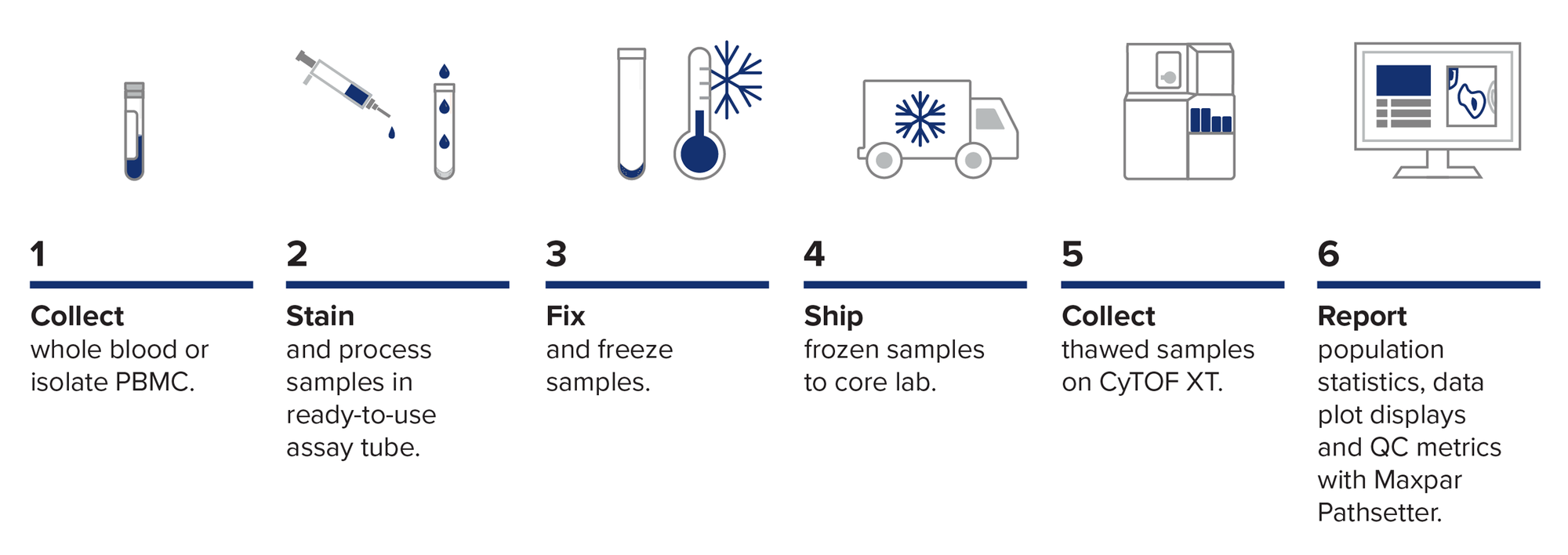
Samples stained with the Maxpar Direct Immune Profiling Assay can be fixed, frozen and stored for analysis at a later time or at another location.
Featured Application
CAR Therapy
With over 2,000 publications and 200 clinical trials, CyTOF® flow cytometry and tissue imaging can be used for a variety of immune and non-immune related diseases. Here we narrow in on an emerging area for high-parameter cytometry.
Chimeric antigen receptor (CAR) cell therapy is a promising breakthrough to significantly improve the management and treatment of cancer. Yet there is much to understand about resistance, toxicity, limited CAR T cell expansion and inadequate persistence in order to gain a stronger foothold in the clinic.
Going beyond the limitations of fluorescence, CyTOF flow cytometry enables simultaneous characterization of CAR cell phenotyping and immune response in a single assay for highly reliable and reproducible results essential for longitudinal, multicenter and clinical trial studies.
Proven performance and reproducibility
The reproducibility of the Maxpar Direct Immune Profiling System has been demonstrated run-to-run and site-to-site.1,2
- Rouphael, N. et al. “Immunophenotyping assessment in a COVID-19 cohort (IMPACC): A prospective longitudinal study.” Science Immunology 6 (2021): eabf3732.
- Bagwell, C.B. et al. “Multi-site reproducibility of a human immunophenotyping assay in whole blood and peripheral blood mononuclear cells preparations using CyTOF® technology coupled with Maxpar® Pathsetter™, an automated data analysis system.” Cytometry Part B Clinical Cytometry 98 (2020): 146–160.
CUSTOMIZATION LETS YOU GO EVEN DEEPER
The Maxpar Direct Immune Profiling Assay was designed with customization in mind.
Choose from 8 ready-to-go 40-plus-marker panel options. Profile T, B, NK and myeloid cells for activation status and cytokine production, all in a single tube.
Building a 47-marker flow cytometry panel to explore antigen-specific immune responses.
Webinars
Reimagine Flow Cytometry
Customization of the Maxpar Direct Immune Profiling Assay and Maxpar Pathsetter for Use in a Biotech Setting
Molecular Characterization of CAR T Cells Using Mass Cytometry
Bringing Immune Profiling One Step Closer to Clinics with Cytometry by Time-of-Flight (CyTOF)

Sasidhar Vemula, PhD
Lead Scientist
Translational Biomarker Solutions
Labcorp Drug Development
Vanderbilt University
Watch now
CyTOF in the Cancer Immune Monitoring and Analysis Centers (CIMAC): Standardization and Harmonization

Holden Maecker, PhD
Professor of Microbiology and Immunology
Director of the Human Immune Monitoring Center Stanford University
A Complete Immune Monitoring Solution with CyTOF: Ideal for Pandemics and Beyond
RELATED BLOG POSTS
- Bringing the CyTOF advantage to CAR cell therapy research
- Using high-parameter flow cytometry and tissue imaging to drive cardiology insights
- Trillions of cells, hundreds of cell types, one collaborative effort
- Five reasons to go beyond the limits of fluorescence with CyTOF technology
- What does high-parameter cytometry really mean?
Resources
Publications
User guides
Technical documents
Customer Stories
Learn more about how our customers are leveraging the Maxpar Direct Immune Profiling Assay.
Lori Turner, PhD

A quick pivot to COVID-19 research with CyTOF
Benjamin Terrier, MD, PhD

Discovering a hallmark phenotype of COVID-19
Don’t have a CyTOF system? We can help!
Maxpar Direct Assays are still the best option for your study if you want convenience, flexibility and consistency.
Maxpar reagents are so robust you can stain, freeze, store and ship your samples for analysis to a CyTOF service provider.
Unless explicitly and expressly stated otherwise, all products are provided for Research Use Only, not for use in diagnostic procedures. Find more information here.









